Article

In Florida, the Florida Boards of Medicine and Osteopathic Medicine adopted a policy for the prescribing of opioids that set the standard of care for the state. The guidelines include performing a patient evaluation before prescribing opioids, obtaining informed consent from the patient for opioid treatment, requiring a written agreement outlining patient responsibilities if the patient is determined to be at high risk for medication abuse or to have a history of substance abuse, conduct a periodic review of the opioid treatment, and maintain a complete medical record of the patient’s treatment.
According to the National Institute on Drug Abuse (NIH), in 2018, nearly 68% of the 4,698 reported drug overdose deaths in Florida involved opioids - a total of 3,189 opioid-related fatalities.
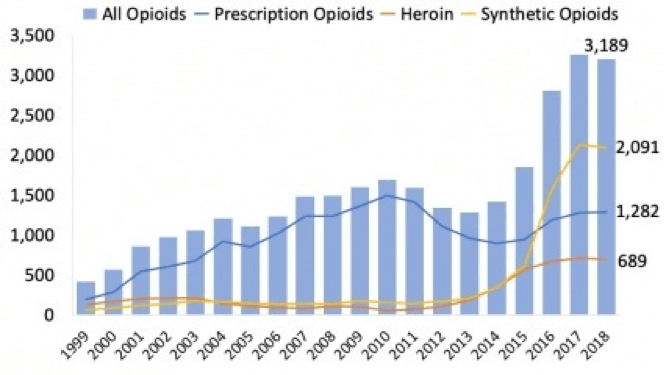
Figure 1. Number of drug and opioid-involved overdose deaths in Florida. Deaths may have involved more than one substance. Source: CDC WONDER, 2020. Source: CDC WONDER.
On July 1, 2018, Florida implemented House Bill 21 (HB21), which restricts the use of prescribed opioids for acute pain to a minimum of 3 days unless specific conditions state otherwise. However, prescribers can extend to a 7-day supply if they deem it medically necessary to do so and document an exception.
Additionally, the restrictions outlined in the bill do not apply to patients suffering from:
The bill also implemented a statewide database, or prescription drug monitoring program (PDMP) requiring action from both pharmacists and providers. PDMP requires pharmacists to report a list of detailed information before filling any prescriptions for controlled substances. Before dispensing these controlled substances, the pharmacist is also expected to consult the database. Failure to do so will lead to several citations resulting in a misdemeanor offense.
Several studies have been conducted on the effectiveness of the HB21 Bill. One such study, the “Changes in Opioid Use After Florida’s Restriction Law for Acute Pain Prescriptions,” was published by researchers at the University of Florida (UF) in February 2020. It analyzed pharmacy prescription claims for opioids dispensed from January 2015 through March 2019 from a single health plan serving more than 45,000 employees of a large Florida employer.
Overall, the study found that the implementation of the HB21 Bill was associated with decreased opioid use and with changes in initial prescribing decisions. As explained by the University of Florida Health, “UF researchers reported that the number of new opioid users per month dropped 16% immediately after the law was implemented, and the number of new users continues to decrease each month. Additionally, the average days’ supply fell from 5.4 days prior to the law to three days.”
The HB21 Bill limits opioid prescribing for primary care, internal medicine physicians, and others who do not specialize in pain management. As noted above, it specifically limits the prescribing of Schedule II opioids for acute pain to 3 days. Exceptions can be made and prescriptions can be extended to a 7-day supply, but prescribers must document “ACUTE PAIN EXCEPTION” on both the patient’s record and prescription.
It is important for providers to remember that this bill did not change how patients are treated for non-acute pain or chronic nonmalignant pain.
Acute pain does not include: (1) cancer, (2) a terminal condition, (3) pain treated with palliative care, (4) a traumatic injury with an injury severity score of 9 or higher, or (5) chronic nonmalignant pain.
The law also requires prescribers to check a patient’s identity and history in the Florida Prescription Drug Monitoring Program (E-FORSCE) before writing each prescription. Failure to consult E-FORSCE will result in a non-disciplinary citation to the prescriber. Any further offences will subject the prescriber to their respective board. It is a first-degree misdemeanor to knowingly fail to report the dispensing of a controlled substance.
Additionally, every DEA-registered provider must take a board-approved, two-hour continuing education course as part of licensing in order to continue prescribing controlled substances.
Though the HB21 Bill only applies to acute pain patients and not chronic pain patients, such as cancer or other forms of trauma, it has been argued that the bill is not well-defined in terms of diagnosis and confusion still exists among prescribers. As noted in the UF study, “There are substantial knowledge gaps around appropriate and inappropriate opioid prescribing, including deficits in understanding current research, legislation, and appropriate prescribing practices. Providers often have knowledge deficits that include:
With a long history of misunderstanding, poor society, provider education, and inconsistent laws, the prescription of opioids has resulted in significant societal challenges that will only resolve with significant education and training.
We encourage providers regardless of state, law, rule or medical guidelines to consider the following items when prescribing opioids in the state of Florida:
These monitoring activities, as well as others, should always be combined with the provider’s thorough assessment of the patient in order to achieve the best outcomes.
Learn more about these recommendations by reading:
Best Practices to Prevent the Misuse of Prescription Drugs
When it comes to the five items to consider listed above, one item stands out among the rest. Toxicology screening is clearly the most objective and scientific way to detect and prevent opioid misuse. Toxicology testing allows providers to intervene at the first sign of medication misuse, thereby preventing continued misuse eventually leading to a use disorder.
DRUGSCAN is a national toxicology testing laboratory that partners with organizations and providers for healthier lives and safer workplaces. Our team takes a proactive approach to help providers in large institutions and smaller private offices alike with the tools necessary to safely prescribe and monitor the use of opioids.
Contact us to learn more about our Prescription Drug Monitoring services and how DRUGSCAN can help you make critical decisions regarding your patients, quickly and efficiently.